OxyContin Files
|
After some physicians began prescribing OxyContin more frequently than every 12 hours, Purdue summoned its sales force to special seminars. As this 2001 presentation shows, company officials were concerned more frequent dosing would hurt business. |
|
In a 2002 federal suit, Alabama businessman H. Jerry Bodie accused Purdue of overstating the duration of OxyContin, among other complaints. The lawsuit was dismissed. |
|
In a 2004 petition to the FDA, Connecticut prosecutors described the alarm inside Purdue when some doctors began prescribing OxyContin at more frequent intervals. “These numbers are very scary,” sales reps were told. |
|
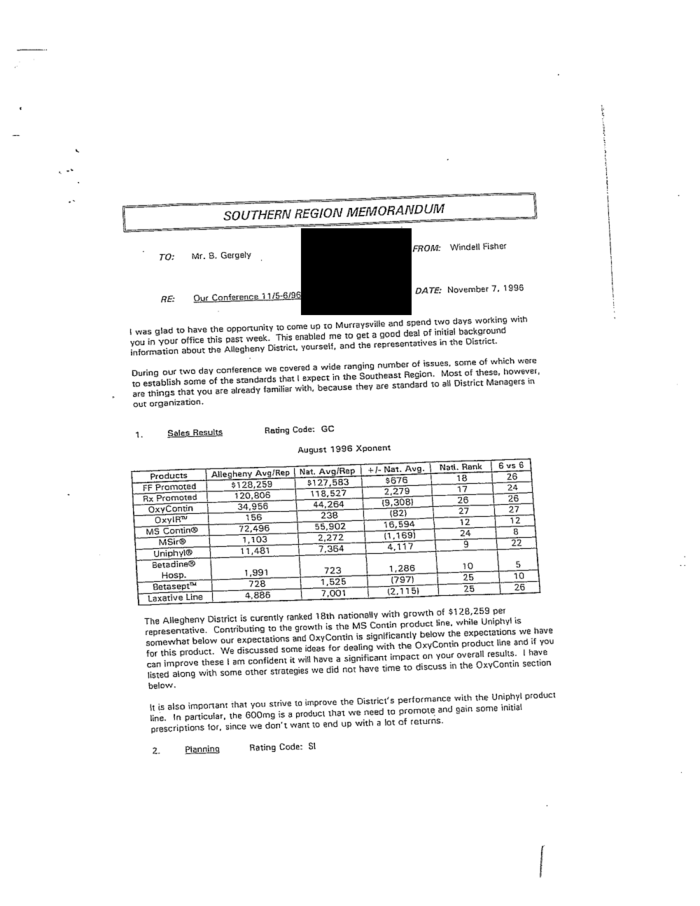
In this 1996 memo entitled “It’s Bonus Time in the Neighborhood,” a Purdue sales manager told her staff to talk up stronger doses of OxyContin in conversations with doctors. |
|
At a 1995 meeting, Purdue executives described how OxyContin could “cure” the “vulnerability” of generic competition and laid out how they planned to market the drug. |
|
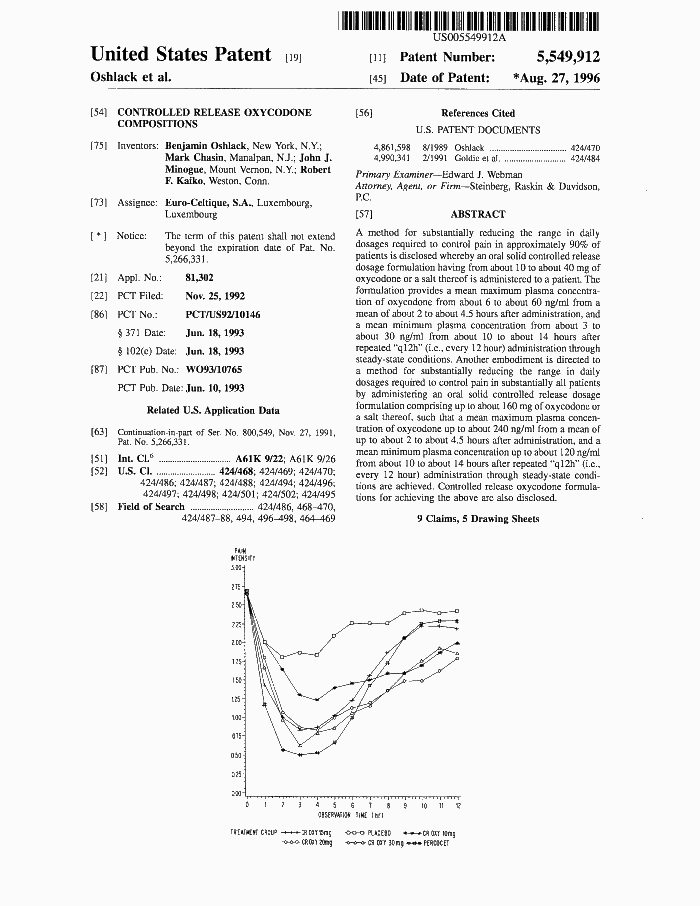
Applying for a patent in 1992, Purdue said OxyContin controlled pain for 12 hours “in approximately 90% of patients.” |
|
When Purdue unveiled OxyContin in 1996, it touted 12-hour duration. |
|
A Memphis doctor testing OxyContin in 1995 allowed half his patients to take the drug three times a day, prompting a recommendation from Purdue’s medical director to prescribe the drug for 12-hour dosing. |
|
In a 2004 letter to the FDA, Purdue lawyers said the company planned to continue promoting OxyContin to doctors as a 12-hour drug for several reasons, including “competitive advantage.” |
|
In this 1990 memo, Robert Kaiko, the scientiest who would go on to help invent OxyContin, explains why Purdue needs another painkiller. |
|
In this 1996 letter, a Purdue regional manager writes that he is concerned about doctors prescribing OxyContin at 8-hour intervals. Sales reps should visit those physicians and convince them to go back to 12-hour dosing, he writes. |
|
In a 2004 decision, a West Virginia judge ruled that Plaintiffs’ shows Purdue “could have tested the safety and efficacy of Oxycontin at eight hours, and could have amended their label, but did not.” Purdue settled the case before trial, agreeing to pay the state $10 million. |